- It was published in The Lancet and corroborated the effectiveness of the vaccine against respiratory syncytial virus.
- But it also showed that those who had to be hospitalized had fewer symptoms than those who were not vaccinated.
Argentina was the first country in the world to include the vaccine against respiratory syncytial virus (RSV) in its national vaccination schedule. Now, a study published in one of the most prestigious scientific journals supports the strategy: the number of babies who had to be hospitalized for acute lower respiratory infections decreased and those who were hospitalized had a better evolution.
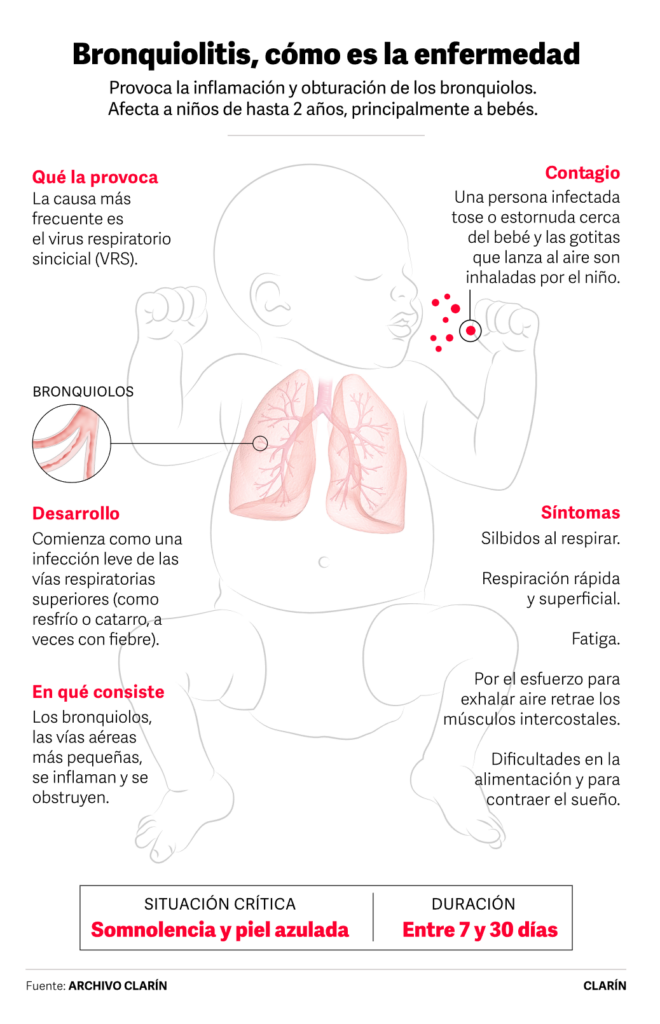
RSV is a virus that is highly transmissible through respiratory secretions or contact with contaminated objects: it is the main cause of bronchiolitis, a disease of which 260,000 cases are reported annually in Argentina in children under two years of age. According to a document issued by the Argentine Pediatrics Society, between one and four out of every 100 healthy infants are at risk of being hospitalized before the age of two.
The disease, historically, was one of the causes of the overloading of pediatric wards every winter, until last year when vaccination of pregnant women began between 32 and 36.6 weeks of gestation, between January and August.
The study published on Monday in the journal The Lancet Infectious Diseases has a long title, but what is important is summarized in its first words: "Real-world effectiveness of vaccination". In other words, what happened to the children of these pregnant women in the first season in which they had to face, outside the maternal uterus, RSV walking around in the street.
The results were very positive, almost within the same parameters as in the clinical trial on which the health authorities based their approval of the vaccine and the preliminary data released at the end of last year.
As now published in The Lancet, the vaccine was 78.6% effective in preventing hospitalizations in infants younger than three months, and 71.3% effective in infants up to six months, the age at which infants are most at risk for RSV complications. In the clinical trial, efficacy was 82.4% at three months and 70% at six months.
"In any vaccine, you need to verify that the efficacy demonstrated in the clinical study is sustained in a large-scale population. To see how the vaccine works, no longer in an ideal setting such as a trial: that it is, for example, equally effective in those who received it at the exact moment or in those who received it a little later", explains the concept of effectiveness (which we heard so much about during the pandemic) Gonzalo Pérez Marc.
The ITRIALS-Equipo Ciencia researcher is one of those responsible for the BERNI study, which was carried out jointly with the Hub for Innovation in Health Policies and Equity of the University of San Martín, the School of Medicine of the National University of La Plata (which is why its authors also include former national civil servants Carla Vizzotti and Analía Rearte) and the laboratory that developed the vaccine. Between April and September last year, 505 babies were studied in 12 public and private health centers in six regions of the country.
For Pérez Marc, these results are very important and consistent with those of the previous study, and support the safety and effectiveness of the vaccine in preventing hospitalizations. But in the exhaustive analysis of the data, another very relevant one emerges: that in the babies who still had to be hospitalized, the evolution of the "vaccinated" ones was better than in those whose mothers had not received the jab.
Although they had enough complications to be hospitalized, the symptoms were milder than in those who had not been vaccinated. For the specialist, it was too small a group to be able to evaluate, for example, how many days of hospitalization were reduced in vaccinated babies, but the results are encouraging. And he emphasizes something else: in the population studied there were three babies who died from respiratory infection caused by the syncytial virus, but none of the mothers had received the vaccine.
Pérez Marc points out that last year -the first year of the campaign- only 62.5% of the target population was vaccinated, so, the doctor is confident that, as vaccination becomes more massive, the overall impact of the vaccine will be more visible. This is also being evaluated and will continue to be studied in 2025 and 2026, with the novelty that it will also be analyzed in Uruguay, which incorporated the vaccine this year.
Prior to its approval, the RSV vaccine was studied in clinical phases in Argentina, in a trial conducted by Perez Marc's Science Team. The same researchers are also continuing to study a pediatric intranasal vaccine. Because, so far, the RSV vaccine is approved for pregnant women (who transmit immunity to their babies) between 32 and 36.6 weeks of gestation, between January and August, to be covered when the virus is circulating.
In addition, for infants whose mothers were not vaccinated, there is the possibility of receiving a monoclonal antibody: there are two on the market, one with free coverage in higher risk cases.
There are some places, such as Chile or Galicia, in Spain, that instead of vaccinating pregnant women, prefer to apply an antibody to newborns. For Pérez Marc, the results of the study will be very useful for decision-makers, not only at the local level to continue supporting the campaign but also so that other countries can analyze the cost-effectiveness of vaccination when adopting a strategy to prevent respiratory syncytial disease.

